What the successful test of a pig-to-human kidney transplant means
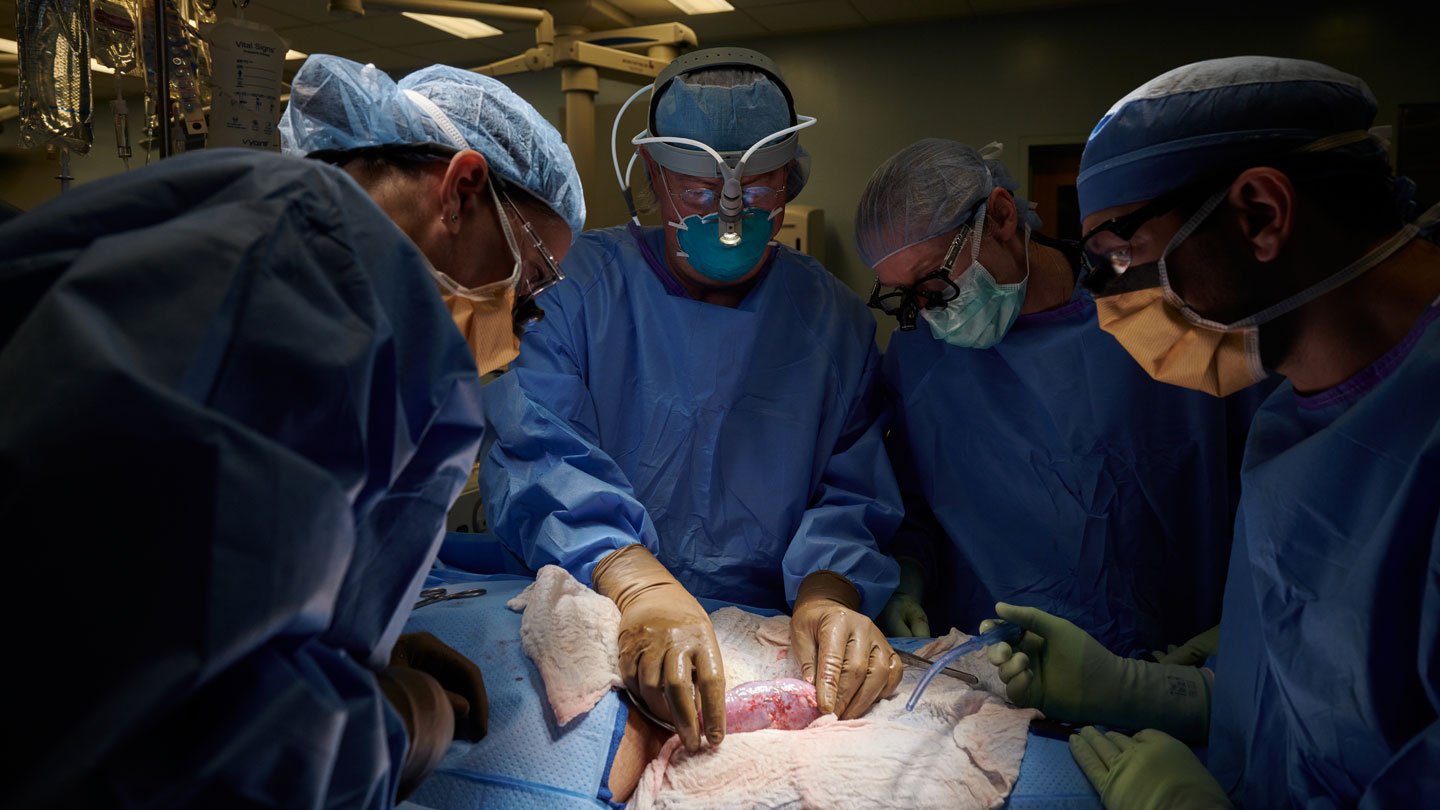
Table of Contents
Surgeons in New York City successfully attached a pig kidney to a human patient and watched the pinkish organ function normally for 54 hours. While such procedures have been done in nonhuman primates, this is the first time that a pig kidney has been transplanted to a human body and not been immediately rejected.
The procedure, announced in a news conference October 21, marks progress toward the goal of drastically expanding the supply of life-saving organs. Millions of people around the world are waiting for donated organs, many of which never come.
While the details of the procedure have not yet been peer reviewed nor published in a journal, “it’s a significant step,” says Megan Sykes, an immunologist at Columbia University who wasn’t involved in the research. But there are many more steps to be taken before patients waiting for a kidney can easily get one from a pig, she says.
Here are answers to some basic questions about the milestone.
Why pigs?
“We’re never going to satisfy the organ shortage problem with human organs,” says John Scandling, a nephrologist at Stanford University who wasn’t involved in this research. “There’s a limit to the number of deceased donor organs that are viable,” and too few donations from living people.
In the United States, over 100,000 people are on the national transplant waiting list, about 90 percent of whom need a kidney. But in 2019, less than 40,000 transplantations occurred. About 17 people die each day while waiting for an organ, according to the Health Resources & Services Administration.
Scientists have long sought to solve this shortage by using animal organs, a field known as xenotransplantation. Pigs have emerged as the primary focus of this research, since their organs are anatomically similar to human organs, and the animals can be bred in a highly controlled manner.
But simply transplanting another species’s organ into a person causes the immune system to revolt at the presence of a large, foreign object. When researchers tried such experiments using nonhuman primates in the mid-20th century, the scientists found that the transplanted organ would quickly turn black. “You could visibly see the organs fail in those days because there’s an immediate reaction,” Scandling says. That immediate reaction, called a hyperacute rejection, is the first major obstacle for a xenotransplant to stick.
In recent decades, genetic engineering has overcome some of these challenges. Scientists learned that the aggressive immune response seen after a pig xenotransplant is spurred on by antibodies that detect a specific sugar molecule called alpha-gal that dots pig blood vessels. It’s the same sugar molecule responsible for some allergic reactions to red meat (SN: 7/30/18).
In the early 2000s, scientists devised ways of disabling the pig gene responsible for this sugar. Subsequent studies have shown that kidneys and other organs from pigs with this gene disabled can be successfully transplanted to nonhuman primates, including baboons (SN: 12/5/18). But until now, a test of this sort of transplant hadn’t been done successfully in people.
What did the researchers do?
Performing a first-of-its-kind operation like this on a living person raises many ethical questions. But in late September, the family of a woman who was brain-dead but kept alive on a ventilator consented to the operation, according to Robert Montgomery, a surgeon at NYU Langone Health in New York City who led the surgical team. She was taken off life support after the procedure.
The woman was an organ donor, but her organs were not suitable for donation. “I want to [acknowledge] how grateful we are to the family of the decedent, who in a time of profound grief, found a way to help their loved one realize her desire to give a gift to humanity at the time of her death,” Montgomery said at the news conference in New York City.
Over two hours, Montgomery and his team carefully attached a kidney from a pig genetically engineered to lack the alpha-gal sugar to blood vessels in the upper leg of the patient. The kidney was kept outside of the body so the team could assess its function in real time. The pig’s thymus gland, which can help educate the immune system to recognize the kidney as part of the body, was also transplanted with the organ to improve its chances of acceptance, Montgomery said. Drugs that suppress the immune system were also administered.
Within minutes, the kidney started producing large amounts of urine and showed other signs of normal functioning. The team kept close tabs on the kidney for 54 hours and saw no signs of rejection. “The pig kidney functioned just like a human kidney transplant,” Montgomery said.
The research team stopped monitoring at 54 hours per guidance from ethics reviewers.
How big of a deal is the transplant?
To scientists who have been working in xenotransplantation, the news is welcome and important but not terribly surprising.
“We were all pretty confident that hyperacute rejection wouldn’t happen,” says Parsia Vagefi, a transplant surgeon at UT Southwestern Medical Center in Dallas. He notes that pig kidneys of the sort used in this procedure have lasted in nonhuman primates for over a year.
“It’s a proof of principle,” Vagefi says, “but it didn’t really move the needle in terms of the science,” though details of how the surgeons did the procedure could provide a clearer picture of how the patient’s immune system responded, for example.
Peter Cowan, an immunologist at the University of Melbourne in Australia agrees. “This is completely as expected, but nevertheless it is an important piece of evidence to support moving to clinical trials of pig-to-human kidney xenotransplantation.”
What’s next for these sort of transplants?
Surviving for 54 hours is a crucial first step, but “nobody needs a kidney for three days,” Vagefi says. Before pig-grown organs can go mainstream, researchers will have to demonstrate that the organs can survive attacks from other players in the immune system and last for months or years in the human body.
For example, over time T-cells, a type of white blood cell, can come to recognize the transplanted organ as foreign and attack. Immunosuppressing drugs can help mitigate these sorts of responses, though the side effects of such drugs, such as susceptibility to infectious diseases, can be a major burden.
Including the pig’s thymus gland, which helps make immune system cells and antibodies, along with the kidney, may help lessen this longer-term rejection, says Kazuhiko Yamada, a surgeon and immunologist at Columbia University who has worked on this method in nonhuman primates for decades. “It’s like a teacher that can educate [the immune system] to not attack the kidney.”
Researchers will also have to show that such transplants are safe in the long-term to get approval from the U.S. Food and Drug Administration, Yamada says. For example, pig organs can contain retroviruses that lie dormant in genes. Some researchers are using the gene-editing technology CRISPR to remove those viruses to potentially improve safety (SN: 8/10/17).
Pigs may be able to provide other organs too, such as hearts or livers, though different organs present their own challenges.
“We’ve been working on this for 20 years,” Yamada says, and now it seems “we’re rounding the final corner and can see the finish line.” But exactly how long it will take before someone in need of a kidney can easily get it from a pig remains to be seen, he says.
Is raising pigs to harvest their organs ethical?
A future where potentially millions of pigs are raised to harvest their organs for humans raises significant ethical questions. “Pigs aren’t spare parts and should never be used as such just because humans are too self-centered to donate their bodies to patients desperate for organ transplants,” the organization People for the Ethical Treatment of Animals, or PETA, said in a statement.
Advocates for xenotransplantation argue the potential benefits of vastly expanding the organ supply are worth any potential harm done to pigs.
“Nearly half of the patients waiting for a transplant become too sick or die before receiving one,” said Montgomery, the NYU surgeon. “The traditional paradigm that someone has to die for someone else to live is never going to keep up with the ever increasing incidence of organ failure.”
What does the first successful test of a pig-to-human kidney transplant mean?







