New technological innovation utilizes everyday sunlight to disinfect drinking drinking water

At minimum 2 billion people worldwide routinely consume h2o contaminated with disease-resulting in microbes.
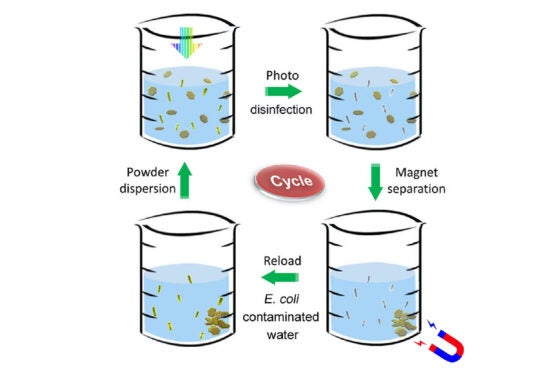
Disinfectant powder is stirred in microbes-contaminated h2o (upper still left). The combination is uncovered to sunlight, which promptly kills all the bacteria (upper correct). A magnet collects the metallic powder soon after disinfection (decreased suitable). The powder is then reloaded into yet another beaker of contaminated h2o, and the disinfection course of action is repeated (reduce remaining). (Image credit history: Tong Wu/Stanford College)
Now, researchers at Stanford College and SLAC Nationwide Accelerator Laboratory have invented a low-price, recyclable powder that kills 1000’s of waterborne microbes for each second when uncovered to everyday sunlight. The discovery of this ultrafast disinfectant could be a significant progress for approximately 30 p.c of the world’s inhabitants with no entry to safe and sound consuming drinking water, according to the Stanford and SLAC group. Their results are printed in a Might 18 study in Mother nature Drinking water.
“Waterborne disorders are responsible for 2 million deaths yearly, the bulk in young children less than the age of 5,” reported research co-direct creator Tong Wu, a former postdoctoral scholar of elements science and engineering (MSE) in the Stanford School of Engineering. “We believe that our novel technological know-how will facilitate groundbreaking alterations in h2o disinfection and encourage more improvements in this exciting interdisciplinary area.”
Typical water-treatment systems incorporate chemical compounds, which can develop harmful byproducts, and ultraviolet mild, which will take a fairly very long time to disinfect and needs a resource of electrical power.
The new disinfectant created at Stanford is a harmless metallic powder that will work by absorbing both UV and superior-strength noticeable gentle from the sunshine. The powder is composed of nano-size flakes of aluminum oxide, molybdenum sulfide, copper, and iron oxide.
“We only employed a tiny amount of money of these resources,” explained senior author Yi Cui, the Fortinet Founders Professor of MSE and of Electrical power Science & Engineering in the Stanford Doerr University of Sustainability. “The products are reduced charge and fairly ample. The vital innovation is that, when immersed in h2o, they all perform collectively.”
Rapid, nontoxic, and recyclable
Soon after absorbing photons from the solar, the molybdenum sulfide/copper catalyst performs like a semiconductor/metal junction, enabling the photons to dislodge electrons. The freed electrons then respond with the bordering water, building hydrogen peroxide and hydroxyl radicals – 1 of the most biologically damaging sorts of oxygen. The newly fashioned chemical substances immediately eliminate the microbes by significantly detrimental their cell membranes.

Microscopic illustrations or photos of E. coli right before (remaining) and immediately after disinfection. The micro organism died speedily after sunlight generated chemicals that brought about critical injury to the bacterial mobile membranes, as revealed in the crimson circles. (Picture credit history: Tong Wu/Stanford University)
For the study, the Stanford and SLAC crew used a 200 milliliter [6.8 ounce] beaker of home-temperature h2o contaminated with about 1 million E. coli microorganisms for every mL [.03 oz.].
“We stirred the powder into the contaminated h2o,” explained co-guide writer Bofei Liu, a former MSE postdoc. “Then we carried out the disinfection test on the Stanford campus in actual daylight, and within 60 seconds no are living micro organism ended up detected.”
The powdery nanoflakes can go all over promptly, make actual physical make contact with with a whole lot of germs and get rid of them speedy, he extra.
The chemical byproducts generated by daylight also dissipate quickly.
“The lifetime of hydrogen peroxide and hydroxy radicals is incredibly brief,” Cui stated. “If they really do not immediately obtain microbes to oxidize, the substances crack down into water and oxygen and are discarded in seconds. So you can consume the h2o suitable away.”
The nontoxic powder is also recyclable. Iron oxide permits the nanoflakes to be removed from drinking water with an normal magnet. In the study, the researchers utilised magnetism to gather the similar powder 30 times to treat 30 different samples of contaminated h2o.
“For hikers and backpackers, I could visualize carrying a small quantity of powder and a smaller magnet,” Cui explained. “During the day you set the powder in drinking water, shake it up a very little bit beneath sunlight and within just a minute you have drinkable water. You use the magnet to consider out the particles for afterwards use.”
The powder could also be beneficial in wastewater treatment method vegetation that currently use UV lamps to disinfect taken care of drinking water, he extra.
“During the working day the plant can use obvious daylight, which would work much quicker than UV and would possibly help you save vitality,” Cui said. “The nanoflakes are quite effortless to make and can be speedily scaled up by the ton.”
The analyze centered on E. coli, which can induce significant gastrointestinal sickness and can even be lifestyle-threatening. The U.S. Environmental Safety Agency has established the maximum contaminant-amount intention for E. coli in consuming drinking water at zero. The Stanford and SLAC staff programs to test the new powder on other waterborne pathogens, which include viruses, protozoa and parasites that also trigger critical health conditions and demise.
Yi Cui is director of the Precourt Institute for Electricity and the Sustainability Accelerator in the Stanford Doerr College of Sustainability. He is also a professor of photon science at SLAC National Accelerator Laboratory. Bofei Liu is now a investigation scientist at EEnotech Inc., a h2o purification spinoff co-founded by Cui. Tong Wu is on the faculty of Tonji University in Shanghai.
Other Stanford co-authors are Harold Y. Hwang, professor of applied physics in the College of Humanities and Sciences and professor of photon science at SLAC, and director of the Stanford Institute for Elements & Vitality Sciences previous engineering postdocs Chong Liu, Jiayu Wan, Feifei Shi, Ankun Yang, Kai Liu and Zhiyi Lu and former engineering PhD learners Jie Zhao and Allen Pei.
Funding for the research was supplied by the U.S. Office of Energy.





